You must be 21 years old or older to purchase tobacco products in Tennessee. Other laws and restrictions may be in place. You should be aware of and compliant with you local regulations.
- Home
- Pipes & Tobacco
- The Humidor
- The Lighter Case
- The Chew Bar
- The Cigarette Stand
- Tobacco Cover Up
- Tobacco Politics
- Smokin' Links
- Contact Us
 Age Verification
Age Verification
GATLINBURLIER
TOBACCONIST
~
Mountain Mall
611 Parkway - D Level
Gatlinburg TN 37738
800.862.2204
MENU
 Our menu
Our menuCYTREL NEWS REPORT
The introduction of tobacco supplements in blends with tobacco will open a new era for modification of tobacco products. While the future potential to alter tobacco appears limited, continuing research on supplements is indicating that advanced variants, already being researched, can produce cigarettes with even greater reduction of smoke components.
For the tobacco manufacturer, a new dimension in product design will be available to meet the changing requirements of the consumer. This briefing has been prepared by (NAMES DELETED FOR LEGAL PURPOSES), Celanese Corporation and (NAME DELETED FOR LEGAL PURPOSES), who have been closely associated in the UK with the technical research and development of Cytrel Tobacco Supplements. It presents a review of tobacco supplements against the background of the industry's final submissions to the Independent Scientific Committee on Smoking and Health.
Official go-ahead awaited on tobacco supplements
These submissions were required under the scientific guidelines announced by the committee, under the Chairmanship of Dr. R. B. Hunter, Vice-Chancellor of Birmingham University, in April 1975.
There are two supplements awaiting approval New Smoking Material, the product jointly developed by Imperial Tobacco and ICI, and Cytrel Tobacco Supplements produced by Celanese Corporation, one of the w orld's leading chemical companies.
Opens a new era
John Offerdahl, Market Director of Cytrel Tobacco Supplements, said: "The official go-ahead for supplements will open up a new era in smoking for the consumer. Cytrel, for example, contains no nicotine and has one-third to one-seventh the tar content of average tobacco. It is cellulosebased, looks like tobacco and takes on the taste of the tobacco with which it is blended."
(NAMES DELETED FOR LEGAL PURPOSES) said: "We examined a range of tobacco supplements including Cytrel. Clearly, the financing of research in this whole area is extremely expensive and the decision to concentrate on Cytrel was taken after careful and extensive examination of the available alternatives."
(NAME DELETED FOR LEGAL PURPOSES) said: "Our company have been closely associated with Cytrel and we plan to blend it into our cigarettes for marketing once cleararJre has been given."
The Celanese programme of research and development of Cytrel—they were the first company in the field, starting in 1957 —has involved an investment well in excess of $20 million. It includes more than 18 years of biological and checnical testing, the setting up of a pilot manufacturing plant and the opening a year ago of a fully commercial plant in Cumberland Maryland. This has been producing and shipping Cytrel since October 1975, and has a capacity of over 20 million Ibs. a year.
Encouraging reaction
"Among the three companies," said Mr. Offerdahl, "we have taken very exacting precautions in the development of Cytrel and products containing Cytrel. During the past 19 years Cytrel has also undergone extensive examination by independent and governmental scientific experts and the results attained suggest identifiable opportunities to smokers in terms of lower tar and nicotine content as well as reductions in other smoke components."
Cigarettes containing Cytrel have been on sale on the Continent for two years. They were first launched in Germany by (NAMES DELETED FOR LEGAL PURPOSES) and by British American Tobacco GmbH in Leichte Classe. Public reaction was disappointing, however a short while later in early 1975, the Swiss company, (NAMES DELETED FOR LEGAL PURPOSES), introduced (NAMES DELETED FOR LEGAL PURPOSES) blended with 20 per cent.
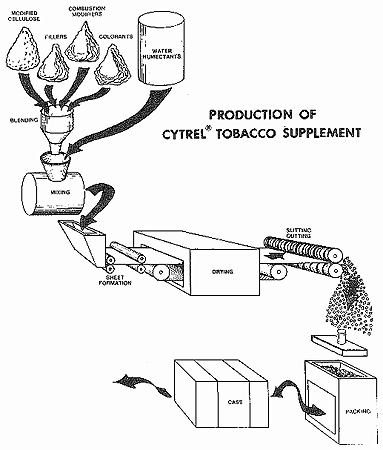
Cytrel Tobacco Supplements
During 1974 both (NAMES DELETED FOR LEGAL PURPOSES) were given approval by the Hunter committee to conduct consumer acceptability tests in the UK with cigarettes containing,blends of Cytrel and tobacco. These tests were held in various parts of the country and the companies report public reaction as "extremely encouraging."
"These tests prove that the British smoker would accept cigarettes containing a blend of Cytrel and we are confident that will be the case when welaunch hopefully soon."
THE HISTORY of tobacco supplements can be traced back sixty years or more when the Germans smoked lime flowers during the first World War. Since then, some manufacturers have used plants, other than tobacco, for various commercial reasons and even some entrepreneurs have turned to things like lettuce leaves. But all have been unsuccessful.
Serious scientific research began in the 1950s in view of the possible interest aroused by articles on tobacco tars which advocated for the first time the production of cigarettes with lower tar yields. Vast sums of industry money were diverted to research in this field, and within five years or so the filter tip was the cigarette on everyone's lips.
Celanese Corporation became one of the leaders in the development of cellulose acetate fibres for filter tips and nearly 20 years ago their scientists turned their attention to a man-made supplement.
1957-58
1958: Celanese funds tobacco supplement research programme.
1960-64
1964:Publication of the first US Surgeon General report on smoking and health. Celanese begins tobacco trade sampling of its prototype tobacco supplement. Celanese broadens first big-systems assay and smoke chemistry analysis.
1965-66
1966: Emphasis on Celanese tobacco supplement programmes moves to product development levels.
1967
Third British company, Courtaulds Ltd., entered the field and commenced research and development on a supplement.called Tabrelle.
An American company—Bravo Inc. of Hereford, Texas—marketed a lettuce leaf cigarette. Initially, they were marketed through the Tesco supermarket chain in Britain. They were subsequently withdrawn due, it is believed, to lack of public acceptance of the taste.
1968
1969-70
1970
1971
1973
British Government appoint Dr. R. B. Hunter, Vice Chancellor of Birmingham University to Chair an Independent Scientific Committee on Smoking and Health. Known as the Hunter Committee.
Courtaulds test-marketed a 50/50 tobacco supplement blend in Coventry and Nuneaton. There was considerable press comment, questions were raised in Parliament and the company was requested to withdraw the brand after three weeks.
Imperial Tobacco announced intention to produce cigarette with a cellulose tobacco supplement. Construction of a £10 million plant for Imperial Developments began in Ardeer, Scotland.
1974
Imperial Tobacco announce plans to conduct consumer acceptability tests in the UK with cigarettes containing NSM. (NAMES DELETED FOR LEGAL PURPOSES) also announced tests with Cytrel. The Hunter Committee had given both companies the go-ahead for these consumer trials.
In Germany, Federal Ministry of Health approve sales of cigarettes containing tobacco s u p p I e m e n t s . First brands launched by (NAMES DELETED FOR LEGAL PURPOSES) The brands, (NAMES DELETED FOR LEGAL PURPOSES) respectively contained blends of Cytrel and tobacco.
Two other German companies, Bayer the chemicals combine and the Reetsma tobacco group announced a joint supplement product, called RCN.
1975
The joint Shaw-Scottish Co-op project in Scotland abandoned.
(NAMES DELETED FOR LEGAL PURPOSES) conduct consumer acceptability tests in the UK with cigarettes blended with several supplements including NSM and Cytrel and decide on Cytrel.
The Hunter Committee three-stage guidelines on the use of tobacco supplements published.
Dr. Gio B. Gori of the National Institure of Health writing in the Journal of the American Medical Association stated that the results of tobacco substitutes may parallel those achieved when manufacturers switched to filters.
"World Tobacco" magazine disclosed that another supplement, the Swiss-made Polystrep, being tested in Europe and a major chemical and fibre company in Japan working with the Japan Tobacco Corporation on a supplement. British American Tobacco also had developed a supplement, called BAT-Flake.
Cytrel plant started world's first commercial production of a tobacco supplement. Annual capacity increased to more than 20 million lb.
1976
Dr. Hunter addressing doctors in Edinburgh about his committee's studies of tobacco supplements said "We have very nearly excluded the possibility of unexpected hazard."
Courtaulds decided to shelve its work on Tabrelle. So did Bayer-Reetsma with RCN. Both groups said they were ready to re-start when the demand for supplements had grown.
In the US, R. J. Reynolds, one of the world's largest tobacco companies, disclosed it had developed a non-tobacco smoking material made from cereal grains that may offer a way to lower cigarette tar and nicotine levels without reducing "smoker satisfaction".
The Polystrep project was discontinued and later acquired by Swiss subsidiary of Phillip Morris.
1977
David Ennals, Social Services Secretary, announced in Parliament a three-year agreement with the tobacco industry which included a stronger on-pack health warning and elimination of high tar brand advertising. Mr. Ennals also announced voluntary agreement with the industry over the testing and marketing of cigarettes containing tobacco substitutes and additives as laid down by the guidelines of the Hunter Committee.


